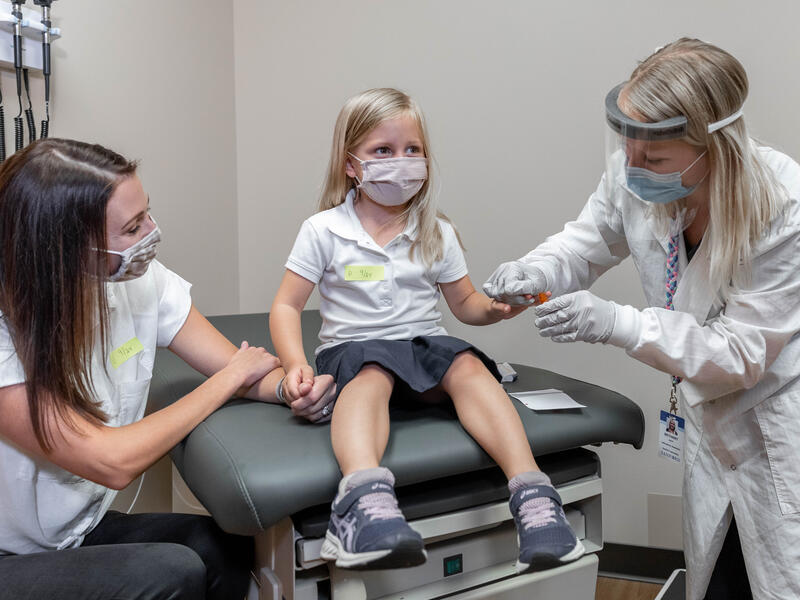
Nearly a century after insulin was first used to treat type 1 diabetes (T1D), it remains the only approved treatment for the disease.
At Sanford Health, efforts are in place to change that. To do so, families are encouraged to enroll children in a screening research study that could lead to identifying T1D before symptoms emerge.
Early detection would allow early treatment and could lead to improved long-term outcomes for both T1D and celiac disease.
Learn more and enroll: PLEDGE pediatric screening study
By participating in the study, families can help develop better ways of identifying children who are at higher risk of developing T1D.
“It was a really easy process,” said Maren Jensen, a Sanford Health content marketing manager whose daughter is enrolled in the study. “I think we’ll do anything we can do to inform our researchers and to help improve treatments or eradicate the disease.”
Jensen’s father was diagnosed with diabetes when he was 9. Because of that, she’s very familiar with the disease from a personal perspective. It motivated her to do what she could to look out for her daughter’s welfare while also contributing to research that could advance treatment of the disease.
Important to make an impact
“It’s super-important to do anything that you can to inform yourself on the future of your child’s health,” Jensen said. “I think that goes for type 1 specifically for me personally because my dad has type 1. It’s important to feel like I’m doing something to make an impact on those who might suffer from type 1 in the future.”
Families of children found to have the antibodies related to T1D are offered:
- Education, monitoring and early intervention to prevent serious illness
- Participation in T1D prevention clinical trials that may delay or stop the progression of T1D
- Referral to clinical care for autoantibodies related to celiac disease
Parents who are 18 years and older can consent for their children to join the study if their child is:
- Between birth and 5 years
- Receiving routine care at Sanford Health
- Not diagnosed with T1D
Depending on a child’s age when entering the study, a small amount of blood will be collected during the course of routine care at birth, at age 2 and/or at age 5. Up until a child’s seventh birthday, a Sanford research team will send a yearly My Sanford Chart invitation to update family medical history. Families will also be invited to complete a short questionnaire about concerns they have about a child developing T1D.
“The fact that Sanford Research is focusing on type 1 in our own backyard, within our footprint, is so incredibly impressive,” Jensen said. “I think we should do anything we can do to equip them with the knowledge that they need to further develop that research. It’s super easy and basically a painless process for us to be able to give back to that research team.”
Learn more
- Sanford study to ID kids’ risk for diabetes, celiac disease
- Artificial intelligence helps spot diabetes risk
- Finding a pediatrician: How to choose care for your child
…
Posted In Endocrinology, Family Medicine, Research